Introduction
Breast cancer remains the leading cause of cancer-related death among women in Europe1, despite recent declines in mortality rates. Over the last 5 years, most European countries have reported significant reductions in breast cancer mortality2, mainly attributed to advances in systemic treatment and, to a lesser extent, improvement in screening and diagnostic methods.
Neoadjuvant chemotherapy has emerged as a key component of breast cancer treatment. Among neoadjuvant strategies, dose-dense chemotherapy regimens, such as those combining doxorubicin and cyclophosphamide (ddAC), have demonstrated superior disease-free and overall survival compared to conventional schedules3. These regimens are administered every 2 weeks with growth factor support and routinely include corticosteroids for antiemetic prophylaxis. Similarly, taxane-based regimens, such as paclitaxel or docetaxel, require corticosteroids in the pre-medication. However, corticosteroid use, particularly at high cumulative doses, has raised concerns regarding immunosuppression and infection risks.
Pneumocystis jirovecii pneumonia (PJP) is a life-threatening opportunistic infection typically affecting immunocompromised individuals. Historically, PJP was commonly associated with HIV infection and hematological malignancies, but its incidence in non-HIV populations, including patients with solid tumors, is increasing due to the growing use of immunosuppressive therapies and corticosteroids4–6. Several reports have described PJP cases in breast cancer patients receiving dose-dense chemotherapy, often occurring between the third and fourth cycles8–11. Waks et al. reported a 0.6% incidence of PJP in patients undergoing ddAC, emphasizing the potential role of corticosteroid exposure and lymphopenia in disease pathogenesis11.
Here, we present three cases of PJP in patients with early-stage breast cancer treated with ddAC followed by paclitaxel chemotherapy at our institution. We aim to heighten awareness of this infectious complication among breast cancer patients and highlight the role of cumulative dosage of prophylactic corticosteroids as a potential risk factor in this clinical setting.
Patient #1
A 46-year-old premenopausal female with no significant past medical history was diagnosed with invasive lobular carcinoma of the right breast (pT2mN1(sn)M0, Luminal B-like subtype). After lumpectomy and sentinel lymph node dissection, she received adjuvant chemotherapy with ddAC every 2 weeks, with pegfilgrastim support, followed by weekly paclitaxel. Antiemetic prophylaxis included dexamethasone 12 mg on day 1, followed by 4 mg twice daily for the subsequent 3 days during ddAC cycles and dexamethasone 20 mg before each weekly paclitaxel infusion.
She completed four cycles of ddAC with good tolerance. Two weeks after completing the last ddAC cycle, before initiating paclitaxel, the patient developed fever (38°C) without focal signs of infection and with a neutrophil count of 4.450/µL and a lymphocyte count of 270/µL. Empiric treatment with amoxicillin/clavulanic acid was initiated for a suspected urinary tract infection (UTI) and proceeded with the first cycle of paclitaxel 24 h later. After being afebrile for 72 h, the fever and generalized malaise recurred, leading to escalation of antibiotic therapy to ciprofloxacin, after a urine culture identified Escherichia coli resistant to amoxicillin.
The patient maintained daily febrile episodes in the following 7 days. Laboratory findings documented no neutropenia (3.360 neutrophils/uL), lymphopenia (380 lymphocytes/μL), and elevated inflammatory markers (C-reactive protein [CRP] 21 mg/dL). Her condition worsened over the subsequent 48 h with progressive dry cough, asthenia, and fatigue. Physical examination revealed tachypnea, decreased bibasilar lung sounds, and severe hypoxemia (partial pressure of oxygen 40 mmHg). Empiric treatment with piperacillin-tazobactam was initiated, and the patient was admitted to the intensive care unit (ICU).
A contrast-enhanced thoracic computed tomography (CT) scan revealed diffuse ground-glass opacities, predominantly in the middle and lower lung lobes and, in a perihilar distribution, multiple areas of established consolidation (Fig. 1). Testing for respiratory viruses such as SARS-CoV-2, Influenza A/B, and respiratory syncytial virus was negative. A flexible bronchoscopy was performed, and P. jirovecii was isolated in the bronchoalveolar lavage fluid (BAL). She was started on trimethoprim-sulfamethoxazole (TMP-SMZ) and methylprednisolone, with progressive clinical, laboratory, and radiological improvement.
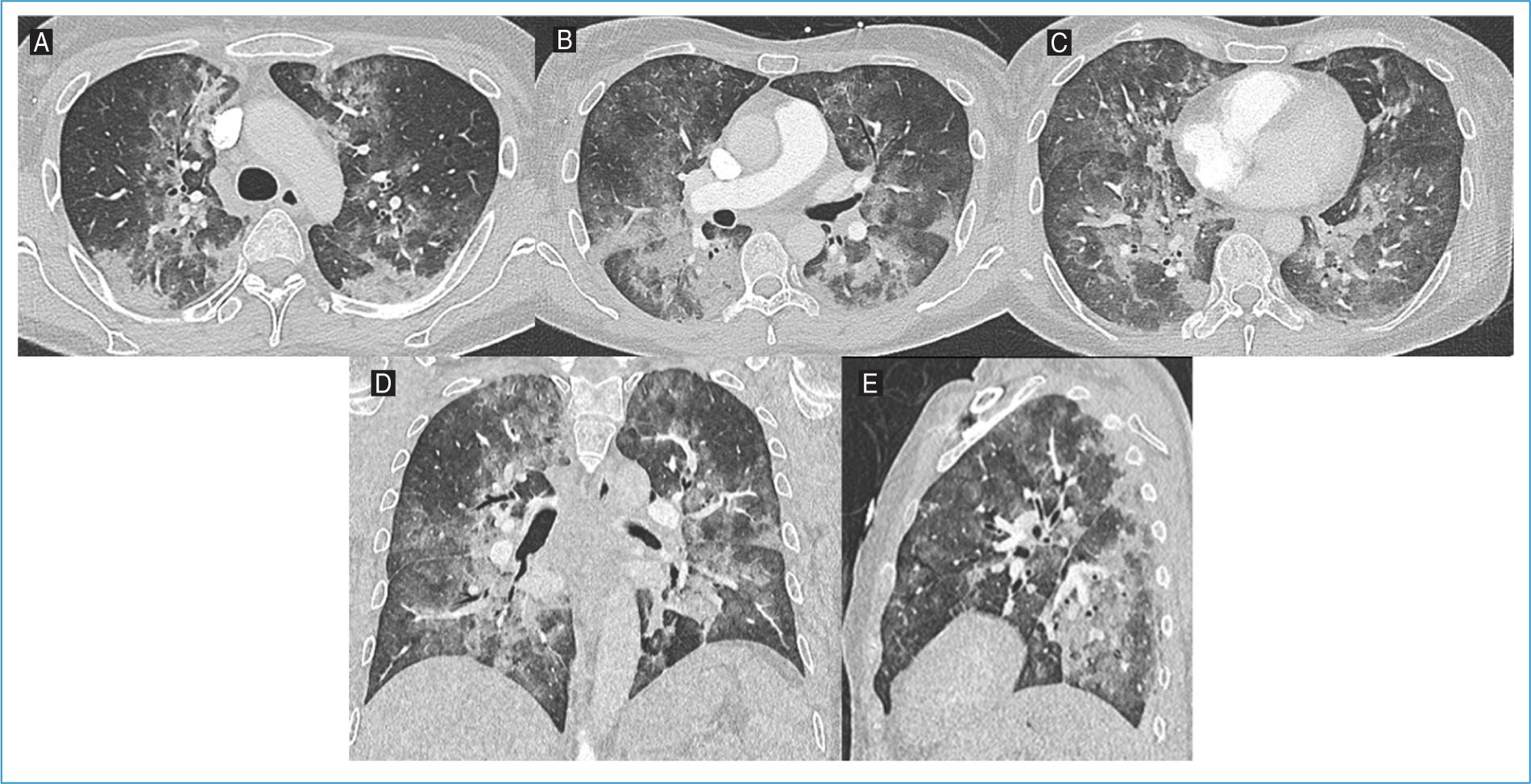
Figure 1. Axial lung window images of the A: upper, B: middle, and C: lower levels, along with D: coronal and E: sagittal reconstructions from thoracic computed tomography angiography of Patient #1. Diffuse ground-glass opacities are observed, predominantly in the middle and lower lung zones and in a perihilar distribution, with multiple areas of consolidation.
Upon recovery, the patient resumed adjuvant paclitaxel without further complications, completing paclitaxel adjuvant treatment successfully. Radiotherapy was subsequently administered, and she has been on hormonal therapy for 18 months without recurrence or further complications.
Patient #2
A 47-year-old female with a past medical history of migraine was diagnosed with left-sided invasive breast carcinoma (cT2N0M0, estrogen receptor and progesterone receptor negative, human epidermal growth factor receptor 2 [HER2] immunohistochemistry [IHC] 3+). She received neoadjuvant chemotherapy with ddAC, followed by paclitaxel, trastuzumab, and pertuzumab. The anti-emesis prophylaxis regimen was identical to Patient #1.
Four cycles of ddAC were administered without toxicity. One week after the first cycle of paclitaxel, she presented with fever and myalgia, prompting a visit to the emergency department. Laboratory tests revealed a neutrophil count of 4000/μL, lymphopenia of 480 lymphocytes/μL, an elevated CRP level (7 mg/dL), and urinalysis with leukocyturia. An UTI was suspected, and she was discharged on cefuroxime. Subsequent urine culture, however, yielded negative results.
One week later, the patient continued with a fever and developed a dry cough and dyspnea. Laboratory tests documented lymphopenia (730 lymphocytes/μL) and an increase in CRP to 27 mg/dL. Chest X-ray documented bilateral basal consolidation. She was admitted to the hospital and started on piperacillin-tazobactam.
During hospital stay, there was worsening hypoxemia with increasing oxygen support and admission to the ICU. Thoracic CT angiography excluded pulmonary embolism and documented extensive pulmonary parenchymal involvement with ground-glass opacities and consolidations, sparing only a few areas in the upper lung lobes (Fig. 2). Tests for respiratory viruses (such as SARS-CoV-2, influenza A/B, and respiratory syncytial virus) and Legionella pneumophila and Streptococcus pneumoniae antigens were negative.
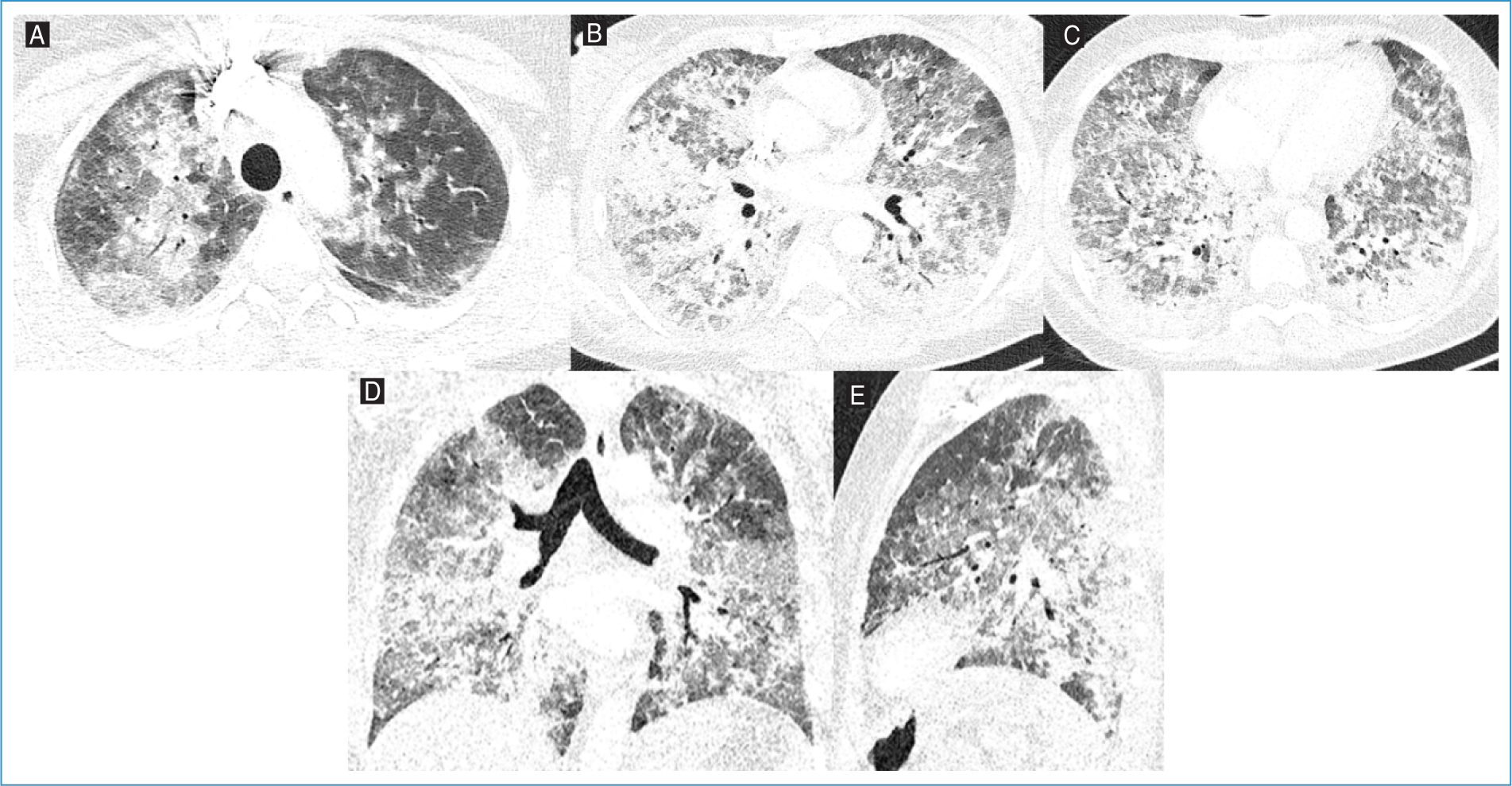
Figure 2. Axial lung window images of the A: upper, B: middle, and C: lower lobes, along with D: coronal and E: sagittal reconstructions from thoracic computed tomography angiography of Patient #2. Extensive pulmonary parenchymal involvement with ground-glass opacities and consolidations, sparing only a few areas in the upper lung lobes.
There was worsening respiratory status, with acute respiratory distress syndrome (ARDS), requiring invasive mechanical ventilation. A bronchoscopy with BAL was performed, and empirical treatment with azithromycin, high-dose TMP-SMZ, and prednisolone (40 mg twice daily) was initiated. P. jirovecii was identified in BAL, establishing the diagnosis of PJP, leading to discontinuation of azithromycin and piperacillin-tazobactam.
There was steady clinical improvement, which enabled ventilatory weaning and successful extubation. Following extubation, oxygen therapy was gradually reduced, and she was discharged in stable condition without the need for supplemental oxygen.
Upon recovery, the patient resumed neoadjuvant therapy without further complications, successfully completing the chemotherapy regimen with a pathologic complete response at surgery. She subsequently underwent radiotherapy and has since been on trastuzumab to complete 1 year of treatment along with hormonal therapy, both well well-tolerated without unexpected toxicity.
Patient #3
A 63-year-old female with multiple comorbidities, including peripheral arterial disease (with stent placement 5 years before), hypertension, dyslipidemia, and a heavy smoking history of 66 pack-years, was diagnosed with invasive carcinoma of the left breast. She underwent lumpectomy and axillary lymph node dissection, with a pathological stage pT2N1aM0, Luminal A-like subtype, HER2-low (IHC 2+ in situ hybridization negative) cancer. Adjuvant chemotherapy was recommended – four cycles of ddAC, followed by 12 weeks of paclitaxel. The anti-emesis regimen for the ddAC protocol was the same for Patients #1 and #2, but the dexamethasone dose for paclitaxel pre-medication was reduced to 8 mg in response to knowledge of the previously reported cases.
The patient presented to the emergency department 6 days after her second paclitaxel treatment with a 4-day history of productive cough with white sputum, dyspnea on minimal exertion, fatigue, and fever (38.5°C). Physical examination documented low peripheral blood saturation (SpO2 93%), tachypnea, and bilateral basal crackles. Arterial blood gas analysis confirmed hypoxemia (pO2 41 mmHg). Laboratory investigations demonstrated lymphopenia (470 lymphocytes/μL) and elevated CPR (15 mg/dL) without neutropenia (7360 neutrophils/uL).
Thoracic CT angiography ruled out pulmonary embolism and showed diffuse ground-glass opacities, predominantly in the upper and middle lung lobes, plus reticular opacities and septal thickening, suggestive of an inflammatory/infectious process (Fig. 3). The patient was admitted for further investigation and management. Initial treatment consisted of amoxicillin-clavulanate, azithromycin, and corticosteroids. However, due to rapid respiratory deterioration, the antibiotic regimen was changed to piperacillin-tazobactam within 48 h. Bronchoscopy revealed thick mucus secretions bilaterally, and BAL diagnosed PJP. Empirical antibiotics were de-escalated, and targeted treatment with high-dose TMP-SMZ and corticosteroids was initiated. Additional diagnostic tests, including bacterial cultures, respiratory viral panels, and galactomannan antigen, were negative. Cytological analysis of BAL showed no malignant cells.
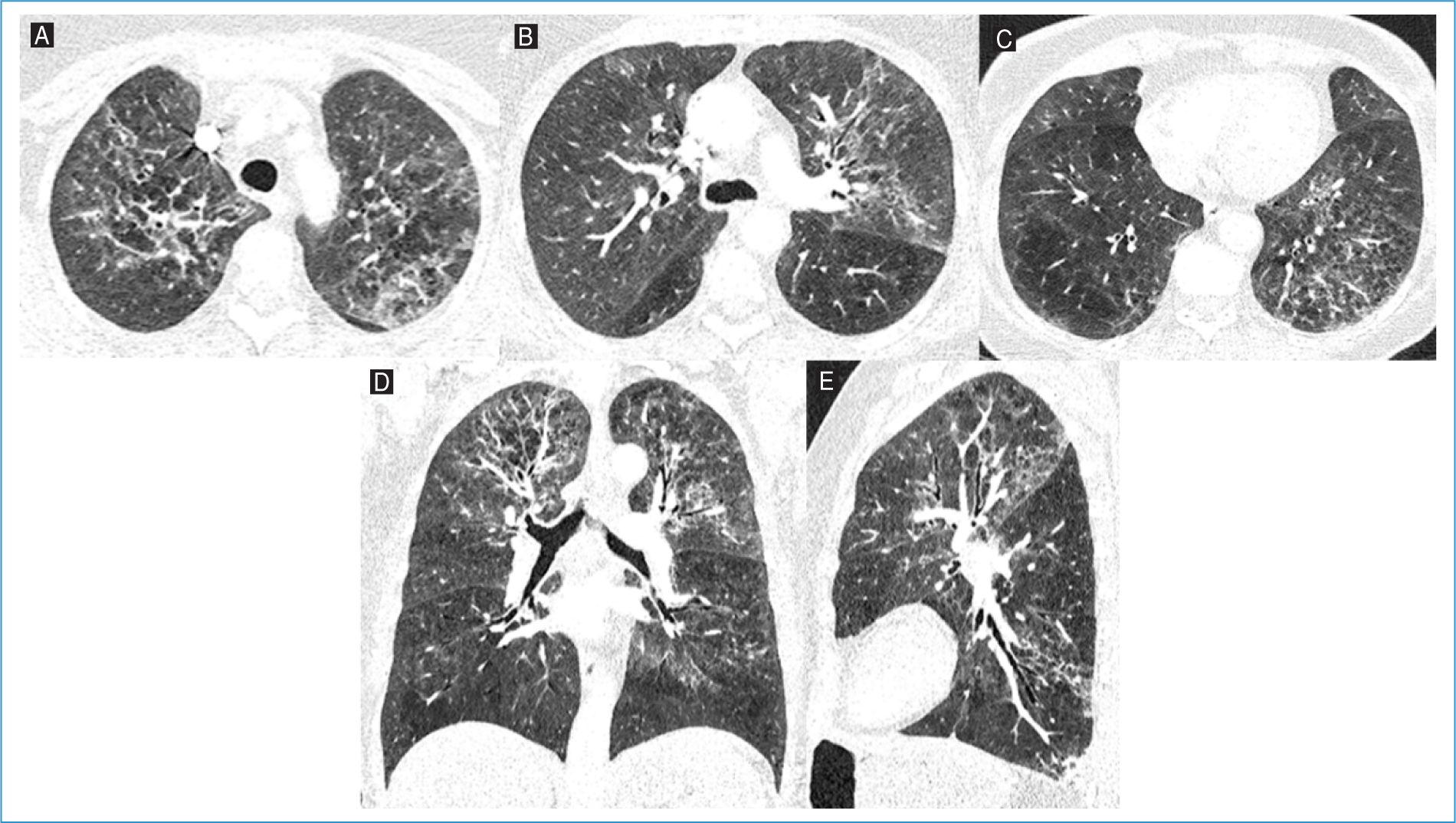
Figure 3. Axial lung window images of the A: upper, B: middle, and C: lower lobes, along with D: coronal and E: sagittal reconstructions from thoracic computed tomography angiography of Patient #3. Diffuse ground-glass opacities, predominantly in the upper and middle lung lobes, are present, accompanied by reticular opacities and septal thickening, suggestive of an inflammatory/infectious process.
The patient improved and was gradually weaned off supplemental oxygen and discharged home in stable condition without the need for further oxygen support. Following recovery, the patient completed adjuvant paclitaxel followed by radiotherapy and has since been on hormonal therapy for 9 months without further toxicities.
Discussion
The three reported cases are consistent with previously reported PJP in patients undergoing dose-dense chemotherapy for breast cancer7,9,12,13. Ours is the second series reported from Portugal8 in this context, stressing the clinical relevance of this treatment complication. Larger population-based studies are essential to accurately assess the incidence and risk factors associated with PJP in this context.
Dose-dense ddAC improves disease-free and overall survival in patients with breast cancer14. Although hematopoietic growth factors, such as pegfilgrastim, promote neutrophil recovery and lower the likelihood of febrile neutropenia, other factors may increase the risk of immunosuppression from intensified chemotherapy.
Lymphocytopenia is a significant contributing factor to the risk of infection with P. jirovecii. Cellular immunity, particularly CD4 lymphocytes, is essential to prevent PJP. Published data indicate that nearly 90% of individuals diagnosed with PJP had lymphocyte counts below 1000 cells/mm3,15. All of our patients exhibited lymphocytopenia below the threshold3.
Another key factor contributing to immunosuppression and PJP is corticosteroid treatment. Patients with breast cancer treated with sequential ddAC and paclitaxel receive corticosteroids over 20 weeks as antiemetics and for paclitaxel hypersensitivity prophylaxis. Corticosteroids are a cornerstone in the prevention and management of chemotherapy-induced nausea and vomiting, offering significant benefits in controlling both acute and delayed symptoms, and are also required for the prevention of infusion and hypersensitivity reactions (HSR) to paclitaxel. However, their immunosuppressive effects warrant consideration, particularly in high cumulative doses.
Intermittent courses of corticosteroids have been previously identified as a significant risk factor for PJP16. In the cases now reported, patients received a total of 144 mg of dexamethasone over four cycles of ddAC administered over 56 days (8 weeks). This translates to an average daily dose of 2.57 mg of dexamethasone, equivalent to 17.1 mg of prednisolone per day based on the dexamethasone-to-prednisolone conversion ratio of 1:6.67, as established in pharmacokinetic studies17. In addition, the first and second cases reported received 20 mg of dexamethasone before each paclitaxel administration, accumulating to a total of 240 mg over 12 weeks. This increased the overall corticosteroid exposure over 20 weeks to a daily equivalent of 18.3 mg of prednisolone. Such exposure exceeds the established threshold for PJP prophylaxis (equivalent of 16-25 mg/day of prednisolone for 4 or more weeks18). The third case received a lower dexamethasone dose (8 mg) as paclitaxel pre-medication. This dosing alteration was made after the evaluation of the first two unexpected cases of PJP and after reviewing with the pharmacy the labeling recommendations of the paclitaxel provider. Although the dexamethasone dose was reduced, the third patient still developed PJP, underscoring the need for caution when using corticosteroids in this setting.
Current guidelines from ASCO19, NCCN, and MASCC/ESMO recommend multimodal antiemetic regimens, incorporating agents with complementary mechanisms of action. Despite overlapping recommendations, slight variations exist between these guidelines17,18. The core recommendations include a 5HT3 receptor antagonist, an NK1 receptor antagonist, dexamethasone, and olanzapine. ASCO and NCCN recommend a three-drug combination of an NK1 receptor antagonist, a 5-hydroxytryptamine type 3 receptor antagonist (5-HT3) receptor antagonist, and dexamethasone for highly emetogenic chemotherapy regimens. MASCC/ESMO advocates for a four-drug regimen incorporating the above three agents plus olanzapine. Regarding dexamethasone pre-medication in breast cancer AC regimens, ASCO and MASCC/ESMO no longer recommend dexamethasone administration on days 2-4 after AC (while NCCN suggests it may be considered), due to limited data supporting dexamethasone use beyond day 1 when an NK1 receptor antagonist is included21,22. These differences highlight the need for a patient-centered approach, where individual risk factors and clinical scenarios guide the choice of antiemetic strategies. By understanding and aligning these recommendations with patient-specific needs, clinicians can optimize antiemetic outcomes while minimizing unnecessary corticosteroid exposure.
Current guidelines emphasize the role of corticosteroids in paclitaxel pre-medication primarily for HSR prevention, rather than for antiemetic purposes. Regulatory-approved prescribing information (RCM)23 recommends dexamethasone doses ranging from 8 to 20 mg before paclitaxel infusion, yet national guidelines and institutional protocols vary in their dosing and duration recommendations. The NHS Northern Cancer Alliance protocol24 recommends 8 mg IV dexamethasone 30-60 min before weekly infusion, aligning with a standardized approach to minimize corticosteroid-related toxicities. Similarly, Clinical Care Ontario25 suggests 10 mg IV dexamethasone before weekly paclitaxel to ensure adequate HSR prophylaxis.
Notably, growing evidence supports reducing or omitting corticosteroid pre-medication in later cycles. A prospective study by Barroso-Sousa et al.26 evaluating a corticosteroid-sparing regimen for dose-dense paclitaxel (biweekly) demonstrated that dexamethasone can be safely omitted in cycles 3 and 4 if no HSRs occur in the first two cycles. This strategy reduced dexamethasone use by 92.8%, with a low overall HSR incidence (4.5%) and < 1% for grade 3/4 reactions in later cycles, supporting the feasibility of a simplified pre-medication approach. These variations highlight a shifting paradigm in corticosteroid pre-medication, moving toward individualized risk stratification rather than routine administration.
The severity of the PJP clinical course varied among the three cases. Patient number 1 required ICU admission for respiratory support, but without the need for invasive ventilation. In contrast, patient number 2 developed ARDS, necessitating invasive mechanical ventilation despite rapid recovery. Patient number 3, despite significant respiratory deterioration, did not progress to ARDS or require intubation. All three patients were able to resume cancer treatment after rapid recovery from PJP.
Our findings, as other published case series7–10,12,13, emphasize the need for heightened awareness of PJP risk in this population. They suggest that corticosteroid exposure in combination with chemotherapy-induced lymphopenia and the underlying malignancy significantly increases susceptibility to opportunistic infections. Consideration should be given to TMP-SMX prophylaxis in patients undergoing dose-dense regimens with high cumulative corticosteroid exposure and lymphocytopenia, as well as evaluating the potential for reducing corticosteroid dosages in chemotherapy protocols to mitigate infection risks.
Our experience with these three PJP cases prompted a revision of our chemotherapy ddAC/paclitaxel protocol, with the reduction of the antiemetic dexamethasone dose currently under review and an adjustment of paclitaxel pre-medication to 8 mg before the first four cycles, followed by a decrease to 4 mg after week 4 if well tolerated.
We acknowledge that this case series is limited by its small sample size but is consistent with published literature. We hope to increase awareness of PJP, an opportunistic infection not usually considered in the setting of (early) breast cancer treatment. Attempts to reduce corticosteroid dosage or consideration of TMP-SMX prophylaxis should be considered when prescribing ddAC paclitaxel chemotherapy.
Conclusion
PJP is a rare but critical infectious complication from chemotherapy treatment of early breast cancer. The combined effect of chemotherapy-induced lymphopenia and corticosteroids for antiemetic prophylaxis and paclitaxel administration requires vigilant monitoring and prompt management. Minimization of preventive steroid dosage or temporary PJP antimicrobial prophylaxis should be considered.
In response to these three PJP cases, adjustments were implemented in our institutional ddAC/paclitaxel chemotherapy protocol. Specifically, dexamethasone pre-medication for paclitaxel was revised to 8 mg during the first four cycles, followed by a reduction to 4 mg from week 5 onward in patients with good tolerance. These adjustments may help guide future clinical practice in balancing effective supportive care with infection risk mitigation. However, further large-scale, population-based studies are essential to better define its true incidence and to identify patients at the highest risk.
Funding
None.
Conflicts of interest
None.
Ethical considerations
Protection of humans and animals. The authors declare that no experiments involving humans or animals were conducted for this research.
Confidentiality, informed consent, and ethical approval. The authors have followed their institution's confidentiality protocols, obtained informed consent from patients, and received approval from the Ethics Committee. The SAGER guidelines were followed according to the nature of the study.
Declaration on the use of artificial intelligence. The authors declare that no generative artificial intelligence was used in the writing of this manuscript.
