Introduction
Nasopharyngeal carcinoma (NPC) is classified into three subtypes: keratinizing squamous cell carcinoma (type 1), non-keratinizing squamous cell carcinoma (type 2), and undifferentiated carcinoma (type 3)1,2. The initial symptoms of NPC may be subtle, with an estimated delay of approximately 6 weeks between symptom onset and the first medical evaluation1. Approximately 70% of patients present with advanced-stage disease at diagnosis1. The gold-standard diagnostic procedure for NPC is nasopharyngeal endoscopy with histological biopsy2.
NPC exhibits unique characteristics, particularly its association with Epstein-Barr virus (EBV) infection, especially in subtypes 2 and 31,2. EBV is classified as a Group 1 carcinogen by the International Agency for Research on Cancer, indicating sufficient evidence of carcinogenicity in humans2. Ninety percent of the global population harbors a persistent, asymptomatic EBV infection. In regions where NPC is endemic, EBV accounts for 95% of NPC incidence and 100% of NPC-related mortality1. The majority of NPC cases occur in East and Southeast Asia (70%), followed by South and Central Asia (6.3%), North Africa (2.6%), and Southern Africa (2.4%)1.
In cases of NPC, EBV detection in histological samples through in situ hybridization is recommended2. Furthermore, a correlation has been established between plasma EBV DNA concentration and disease stage, clinical outcomes, and overall survival1,2. Incorporating this assessment into the tumor, node, metastasis (TNM) staging system appears to enhance its prognostic accuracy1,2.
In this article, we present the case of a male patient diagnosed with NPC and the monitoring of EBV DNA throughout his clinical course.
Case report
A 45-year-old male was referred to our oncology center with a 2-month history of fever, night sweats, and a left cervical mass. Cervical computed tomography (CT) revealed an extensive conglomerate of lymphadenopathy involving levels II, III, IV, and V on the left side, with no evidence of a primary neoplasm. Excisional biopsy of one of the lymph nodes revealed metastatic undifferentiated carcinoma originating from the upper aerodigestive tract.
The patient underwent positron emission tomography-CT (PET-CT), which demonstrated increased uptake in the nasopharynx and the cervical lymph node conglomerate. Nasopharyngeal biopsy confirmed a diagnosis of non-keratinizing undifferentiated NPC, positive for EBV-encoded RNA and negative for p16. Clinical staging was cT1N2M0 (stage III). Plasma EBV DNA was positive (1.4 × 103 copies/mL).
The patient completed three cycles of cisplatin (100 mg/m2) and gemcitabine (1250 mg/m2) every 21 days until October 2022, with a favorable metabolic response on PET-CT and undetectable plasma EBV DNA levels (Fig. 1). Following multidisciplinary discussion, the patient underwent concurrent chemoradiotherapy (CRT), consisting of radiotherapy to the primary tumor and cervical lymph nodes (total dose of 70 Gy in 35 fractions) and cisplatin (100 mg/m2) every 21 days for three cycles, completed in January 2023.
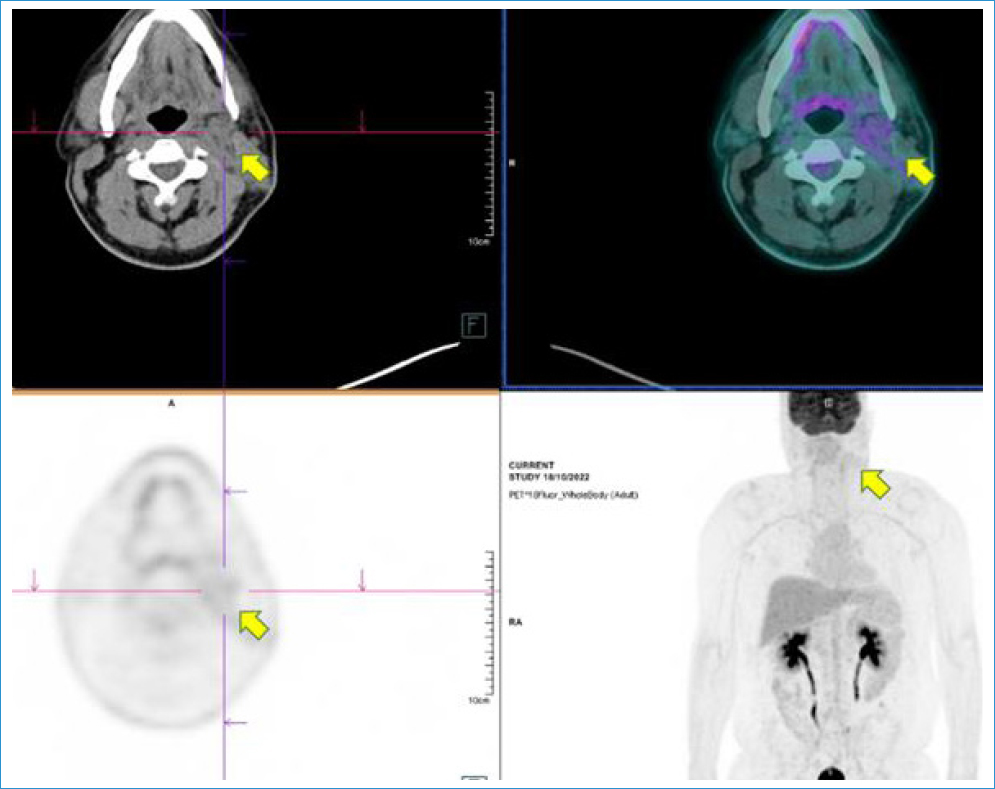
Figure 1. Imaging assessment by positron emission tomography-computed tomography following chemoradiotherapy, showing dimensional and metabolic regression of the left cervical lymph node conglomerate (yellow arrows), compared to the baseline scan.
Three months after completing CRT, a new PET-CT revealed bone and lymph node metastases. Lymph node biopsy confirmed metastasis of the previously diagnosed NPC. At this time, plasma EBV DNA was < 250 copies/mL. He initiated chemotherapy with carboplatin area under the curve (AUC 6) and paclitaxel (175 mg/m2), completing six cycles by October 2023. The main toxicity observed was grade 2 sensory peripheral neuropathy (per CTCAE v5.0).
Post-treatment PET-CT showed disease progression with de novo hepatic metastases, and plasma EBV DNA became positive (7.8 × 103 copies/mL). The patient began a new line of treatment with nivolumab (480 mg every 28 days). After the second cycle, the patient was hospitalized for persistent fever and started on broad-spectrum antibiotics. However, no microbiological agents were identified, and a significant increase in plasma EBV DNA copies was observed (1.0 × 106 copies/mL).
Cervico-thoraco-abdominal CT (CTA) revealed disease progression (lymphatic, bone, and hepatic), with no evidence of infectious focus. After discussion within the medical oncology team and with the patient, rechallenge with gemcitabine (1000 mg/m2 on days 1, 8, and 15 every 28 days) was proposed.
Following the first cycle, the patient experienced sustained apyrexia and clinical improvement (reduction in pain and anorexia). After the third cycle, imaging showed a partial response, and there was a decrease in EBV viral load (< 250 copies/mL) (Fig. 2). Due to grade 3 hematologic toxicity (per CTCAE v5.0), the gemcitabine dose was reduced to 750 mg/m2 after the third cycle.
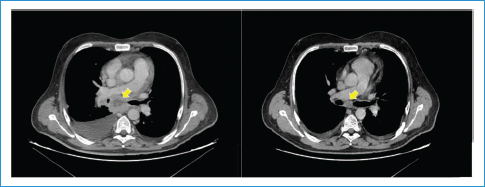
Figure 2. Partial response of lymph node disease (yellow arrow); computed tomography from January 2024 on the left and March 2024 on the right.
After the fifth cycle of treatment, the patient developed a fever and was diagnosed with a severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection. A CTA performed in June 2024 revealed disease progression (lymph nodes and liver). Due to worsening general condition and persistent fever, the patient was hospitalized for further investigation. He was diagnosed with sepsis of undetermined origin and multiorgan dysfunction (respiratory and renal). Despite the therapeutic measures, the patient’s clinical condition continued to deteriorate, and he passed away during hospitalization.
Discussion
The pathogenesis of NPC is influenced by environmental factors (such as tobacco use, alcohol consumption, and diet), host-related factors (including male sex, genetic predisposition, and ethnicity), and EBV infection1.
Between 5% and 15% of patients who undergo radical treatment for NPC will experience locoregional recurrence, and 15-30% will develop distant metastases1. The risk of recurrence following curative-intent radiotherapy follows a bimodal pattern, with peaks at approximately 1.5 and 3.5 years of follow-up2. Plasma EBV DNA detection is thought to enable the early identification of disease recurrence during follow-up (level of evidence V, grade of recommendation B)2. Therefore, plasma EBV DNA quantification should be considered 1-4 weeks after radiotherapy (level of evidence II, grade of recommendation B) and annually thereafter (level of evidence V, grade of recommendation B)2.
In addition, plasma EBV DNA concentration is a prognostic factor for distant metastases in patients eligible for surgical treatment, and both its concentration and clearance rate during therapy are prognostic indicators in patients receiving first-line palliative treatment2.
Immunotherapy is a promising treatment option for unresectable or metastatic NPC, partly due to the high expression of EBV-related antigens and CD4+/CD8+T-cell target proteins3. Immunotherapy may be active (e.g., vaccination, immune checkpoint inhibitors [ICIs]) or passive (e.g., adoptive transfer of autologous EBV-targeting cytotoxic T lymphocytes)3. The most recent update of the European guidelines includes ICIs in combination with chemotherapy as a first-line palliative treatment, whereas ICI monotherapy remains a second-line option4.
Early diagnosis of NPC remains a challenge due to its deep anatomical location and non-specific symptoms1. As such, EBV testing has been investigated as a screening tool in endemic regions and high-risk individuals (particularly men aged 30-59 years), demonstrating utility in detecting early asymptomatic NPC. Even small tumors may release sufficient DNA into the circulation to allow detection5,6.
Serological evaluation of anti-EBV capsid antigen (viral capsid antigen immunoglobulin A [IgA]), anti-EBV nuclear antigen IgA, and early antigen IgA, when combined, shows high sensitivity and specificity (91-96%) for screening purposes, although the positive predictive value (PPV) remains low (< 10%)1,5.
EBV viremia detection has also been studied as a screening method, with high specificity and sensitivity (97% and 99%, respectively), though it is associated with higher costs and the need for advanced laboratory infrastructure5,7. Detection of methylation patterns in circulating DNA appears to be a promising method, given the high PPV reported in studies of other malignancies1,7.
Conclusion
This case report aimed to illustrate how EBV viremia monitoring may be relevant in detecting disease progression or recurrence (Table 1). Thus, monitoring plasma EBV DNA concentration may contribute to a more personalized treatment approach for patients with NPC. We also aimed to highlight the challenges in selecting subsequent lines of therapy following first-line treatment failure in metastatic NPC.
Table 1. Temporal evolution of EBV viral load
| Date | June 2022 | October 2022 | April 2023 | October 2023 | January 2024 | March 2024 | May 2024 |
|---|---|---|---|---|---|---|---|
| Viremia (copies/mL) | 1.40 × 103 | ND | < 250 | 7.80 × 103 | 1.0 × 106 | < 250 | 572 |
| Response evaluation | Baseline | Metabolic response (PET) | DP | DP | DP | PR | NA |
NA: not assessed; ND: not detected; PD: progressive disease; PR: partial response.
Acknowledgment
We would like to thank the Head and Neck Cancer Study Group (GECCP). This work was awarded “Best Tertúlia” and presented at the 14th GECCP Symposium.
Funding
None.
Conflicts of interest
None.
Ethical considerations
Protection of humans and animals. The authors declare that no experiments involving humans or animals were conducted for this research.
Confidentiality, informed consent, and ethical approval. The authors have followed their institution’s confidentiality protocols, obtained informed consent from patients, and received approval from the Ethics Committee. The SAGER guidelines were followed according to the nature of the study.
Declaration on the use of artificial intelligence. The authors declare that no generative artificial intelligence was used in the writing of this manuscript.
