Introduction
Nutrition is critical to the management of cancer, and it is of utmost importance to identify clinical findings that may negatively impact tolerance to treatments, quality of life, and survival, e.g. malnutrition, sarcopenia (weight loss and depletion of muscle mass and function), physical function or cachexia (severe weight loss characterised by a persistent decline in skeletal muscle mass, with or without fat loss)1–4.
Malnutrition affects between 30% and 80% of cancer patients, with the highest prevalence observed in pancreatic (60-80%), gastric (50-75%), oesophageal (60-70%), and head and neck cancers (50-70%), due to tumor location, systemic inflammation, and treatment side effects5. Furthermore, among cancer patients undergoing active treatment, the prevalence of malnutrition or nutritional risk ranges from 40% to 80%, regardless of cancer type, highlighting the need for routine nutritional screening and early intervention as soon as the patients start anti-cancer treatments6.
Loss of muscle mass heightens the risk of dose-limiting toxicity, as depleted patients are less likely to benefit from anticancer treatments7–16. Muscle depletion can directly impair the treatment efficacy, making it essential to identify and assess nutritional risks in all cancer patients, regardless of disease stage12,17,18.
Nutritional factors should be monitored continuously, before, during, and after treatments to enhance efficacy and minimize side effects. Early detection of nutritional impairment is crucial to ensure patients remain eligible for active treatment19–21.
Apart from nutritional parameters, muscle function has emerged as a key marker of sarcopenia and, eventually, cachexia. In head and neck cancer, handgrip strength (HGS) measurement at diagnosis was adequate to discriminate patients with sarcopenia, shorter survival, and those at risk of relapse (Orell et al. 2025; data submitted)22. HGS is an indirect indicator of muscle mass and nutritional status, serving and stands as an important biomarker of health in this patient population. Its utility extends to identifying diverse health issues and its potential as a new vital sign throughout the disease trajectory20,21.
A recent study involving patients with cancer at nutritional risk identified several malnutrition and muscle health parameters as independent prognostic indicators for mortality23. The modified Global Leadership Initiative on Malnutrition, (European Society for Clinical Nutrition and Metabolism [ESPEN]) criteria24, low albumin concentration, reduced HGS, and decreased muscle radiodensity were reported to provide prognostic information regarding 180-day mortality. Again, several parameters reflecting malnutrition and muscle function emerged as independent prognostic indicators of mortality (Orell et al. 2025; data submitted)22.
It is worth noting that nutrition is a fundamental human right that cannot be foreseen or overlooked20. Supporting early diagnosis of muscle loss ensures all patients receive the nutrition care essential for effective treatment. Engaged professionals and easy-to-apply protocols help ensure that all patients using healthcare services receive high-quality nutritional care. Human rights norms, ethical values, and principles can contribute to moving forward in promoting patient’s access to appropriate nutritional care20.
Based on this framework, maintaining an adequate nutritional status and body composition is linked to better tolerance of anticancer therapies, fewer post-surgical complications, and improved long-term outcomes, including survival21,24–30.
Experts agree on the importance of early identification of patients at risk of malnutrition or muscle loss, and recent international guidelines advocate for incorporating nutritional screening, assessment, treatment, and monitoring as integral components of cancer care31–34.
Therefore, a standardized approach to nutritional diagnosis, nutrition intervention, and monitoring is necessary.
Several tools/protocols exist for nutritional screening and stratification, but none integrates muscle function. A quick diagnosis integrating nutritional risk and muscle function seems the most useful and informative in the long term30, and this is what we propose in the present article. It is mandatory to integrate this critical task into the whole process of the patient disease management, ensuring it is feasible and effective within a busy oncology practice34.
Ideally, such a protocol can be integrated into a machine-learning model and swiftly transferred to any institution’s patient management platform.
Objectives
Our main goal is to provide a practical protocol to identify the risk of malnutrition, of muscle mass depletion, and of muscle dysfunction in patients with cancer, which is key information for the treatment plan enabling timely evaluation and follow-up by a multidisciplinary team. Applicable at the start of any anti-cancer therapy, regardless of cancer type or stage, this protocol is straightforward and efficient for regular monitoring and early detection of nutritional issues throughout treatment. With this article, we intend to empower clinicians to swiftly identify at-risk patients, facilitating prompt referral for comprehensive nutritional assessment by a clinical nutritionist. This protocol is not intended to replace existing guidelines or tools for nutritional evaluation but to ensure that patients needing such support are quickly identified and managed. As medicine evolves, we foresee that in a near future, this protocol could be integrated into patients’ records using machine learning, allowing full integration of nutrition information in all medical decisions. It is noteworthy that universal nutritional care greatly benefits patients with cancer, and shared responsibility in identifying, referring to, and intervening nutritionally is the expected quality of all healthcare institutions.
Methods
A multidisciplinary group of national/international experts in oncology, with a specific focus on nutrition, and with medicine, oncology, nutrition, and nursing backgrounds was selected to integrate an expert panel, consisting of two physicians (one KOL in clinical nutrition, metabolism, and oncology), two clinical nutritionists, and an oncology nurse specialist. The panel met periodically to review existing literature, align objectives, and draft the protocol support text monthly. In these meetings consensus was reached through structured discussion, clarification of differing views, and formal agreement, supported by voting or rating methods. There was a final recap meeting in person, with additional discussions via email and Microsoft Teams® as necessary.
The plan was:
– The first meeting listed challenges and potential obstacles perceived by clinical teams regarding nutritional screening and assessment within the patient’s journey.
– After carefully reviewing the literature, the second and third meetings identified key concepts that were used to build the protocol framework, for routine clinical use. Literature review was made by an integrative and narrative review, combining diverse types of evidence to provide context, background, and expert interpretation, to develop comprehensive clinical understanding useful for framing the clinical problem and justifying protocol need, to address complex, multidimensional aspects of care (e.g., nutrition care in cancer).
– Fourth and fifth meetings were used to gather and interpret the evidence generated by the review and consolidate practical recommendations.
– Subsequent meetings were used to discuss and align fundamental components and tools identified, to form the basis of the practical protocol to identify nutritional risk and muscle depletion in oncology and design a reproduceable format of the protocol.
Results and discussion
The nutritional care process, as a systematic approach that aims to provide the best nutritional care, is a process described in four stages: assessment, diagnosis, intervention, and monitoring/nutritional evolution31. This process is triggered by screening, and referral should be based on appropriate tools and methods, assuming a multidisciplinary involvement35. The multidisciplinary team is responsible for ensuring that the patient’s nutritional needs are met and that the best care is provided during the disease journey36. This is aligned with Horizon 2030 strategy (Despacho nº 13227/2023)37 that reinforced earlier legislation (Despacho nº 9984/2023)38 to implement systematic nutritional risk screening (NRS) in cancer in the National Health Service (primary care, hospital, and palliative care).
For outpatients, in the National Plan for Oncological Diseases, screening is advised at each care stage or according to clinical indication. The suggestion is to assess, by 2030, nutritional risk in over 50% of patients within a month before their first consultation, and access to nutritional support by specialized professionals in more than 90% of patients at nutritional risk. Ideally, nutrition consultations for outpatients with cancer and at high risk of malnutrition should take place within 15 days after signaling access to nutritional support and nutritional therapy is now predicted in Portuguese legislation (Portaria nº 82/2025/1)39.
Identification of patients with or at risk of malnutrition and/or muscle depletion
Protocol proposal
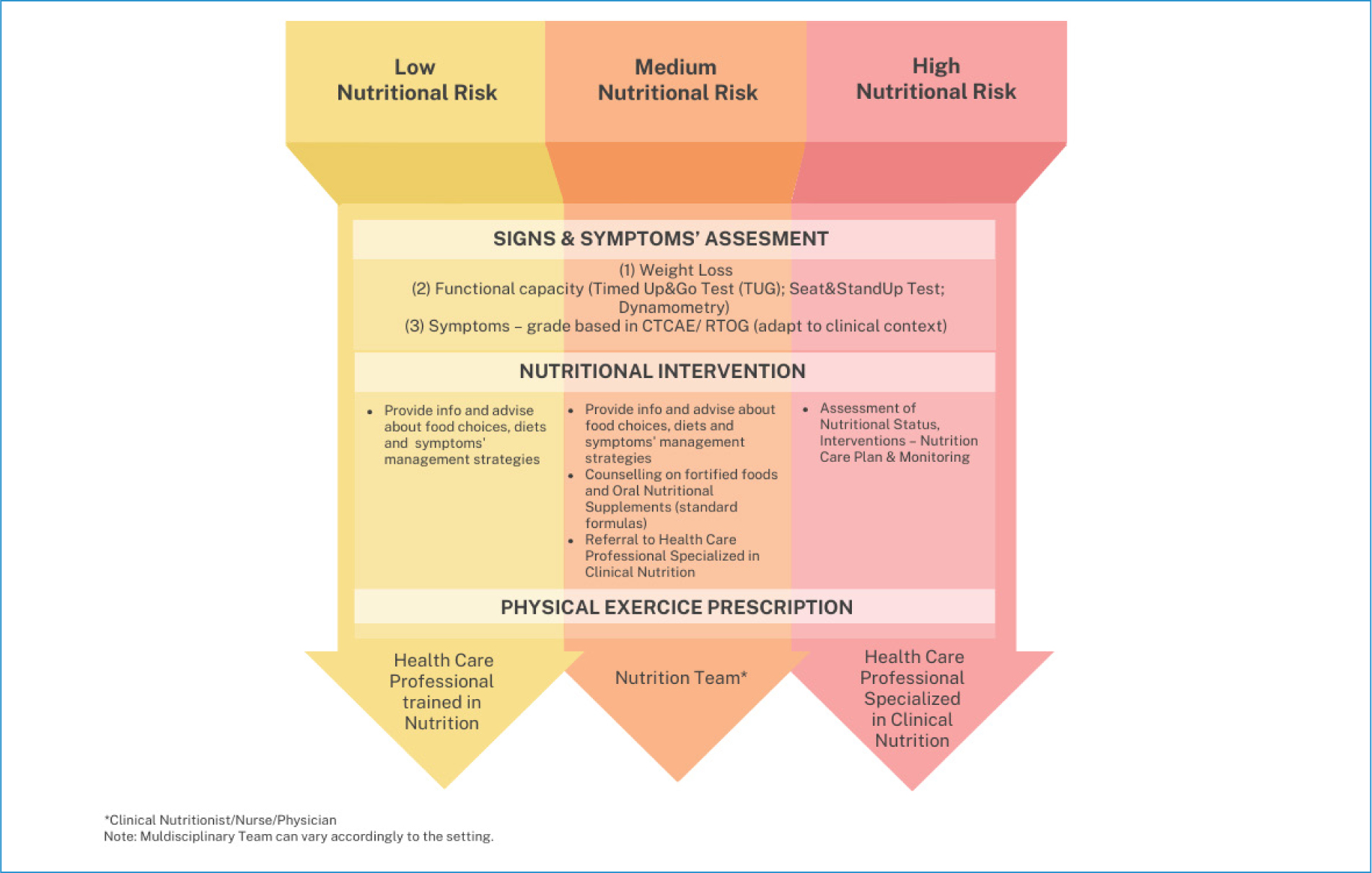
Among the validated tools for nutritional screening, for example, Malnutrition Universal Screening Tool, NRS, NutriScore and Mini Nutritional Assessment, the adequate one should be selected in each institution as the most valid and feasible (Supplementary data I-IV)34–37,40,41; Furthermore, a minimum set of informative questions was identified to determine the risk of muscle depletion and muscle function in patients scheduled to begin antineoplastic therapy and to monitor any change in patients’ nutritional status before and throughout treatments (Supplementary data IV). The expert panel identified factors as essential components to give a rapid understanding of patients’ nutritional and physical status: (1) NRS by validated tool (Supplementary data II): weight history and nutritional intake, and (2) muscle function by dynamometry or test up and go or sit and stand-up test (Supplementary data). Nutritional monitoring and assessment of nutritional risk should be conducted at each treatment cycle, monthly, or as clinically indicated, to ensure timely identification and management of nutritional decline during cancer therapy.
Our proposal is that screening should be used to determine the risk of malnutrition and muscle mass depletion for systematic application36,37. The rapid identification of patients at-risk enables their prompt referral for additional assessment24. All patients with cancer referred for active treatment should be screened, regardless of their tumor type (Fig. 1).
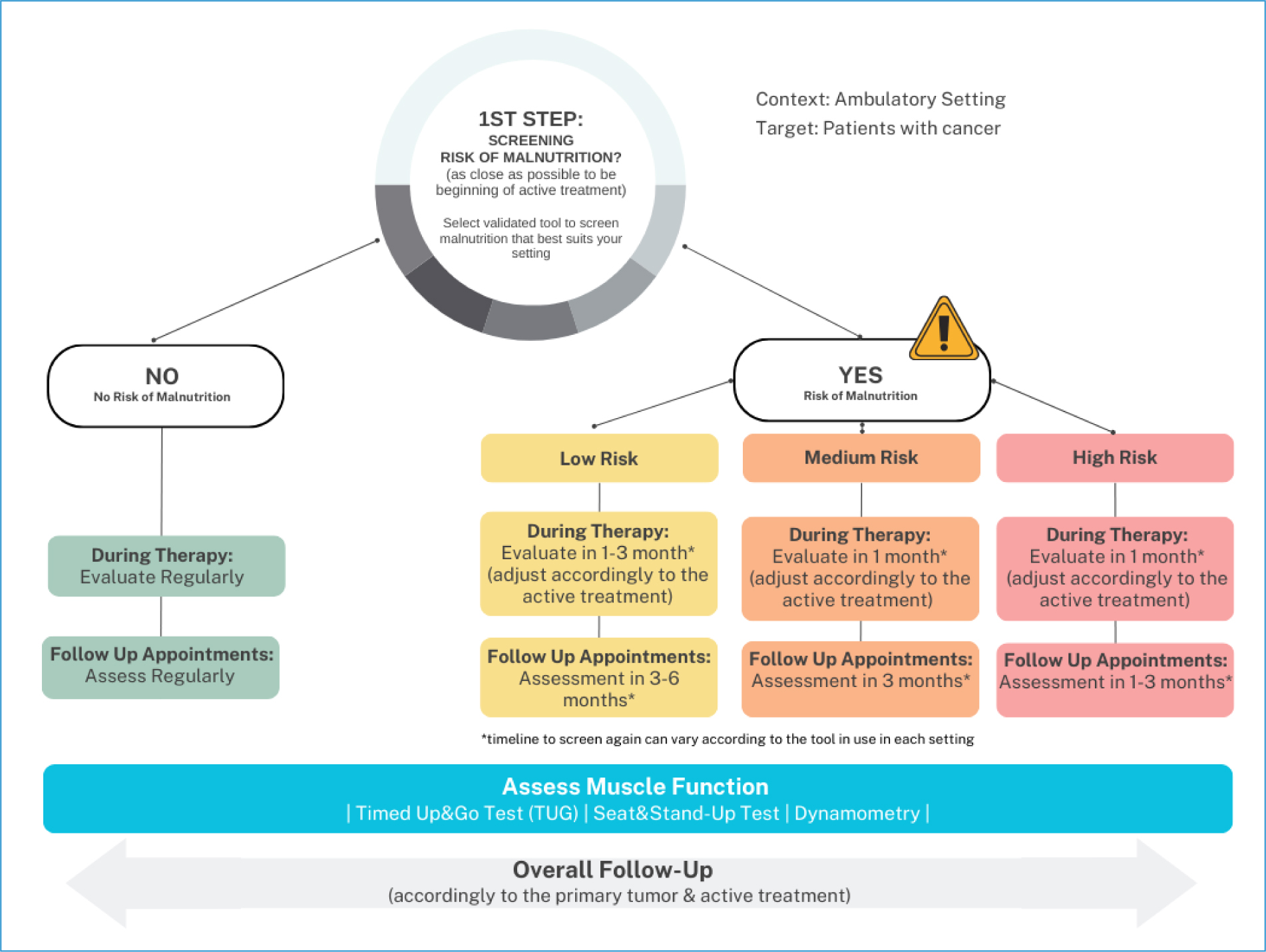
Figure 1. Flowchart for malnutrition and muscle depletion risk assessment.
Screening can be conducted by a nurse, physician, or clinical nutritionist using one of the tools indicated and validated for oncology patients (Supplementary data I-IV)42–47.
Nutritional assessment should occur in patients identified at risk, using validated tools for oncology, ideally as close as possible to the timing of cancer diagnosis. This preliminary information allows for stratifying patients so that the nutritional care process is as efficient and appropriate as possible36.
As muscle depletion is common in oncology patients, it is recommended that, in addition to nutritional screening, muscle mass screening be performed at the time of diagnosis (or as close as possible) and whenever there is a change in the patient’s clinical situation29,48–51.
If the expected survival is 3-6 months, the focus should be on identifying and managing symptoms that negatively interfere with eating and quality of life29.
Reductions in patient-perceived strength and general mobility may be related to muscle depletion, such as their ability to carry out ordinary daily activities (i.e., open bottles, stand up from a chair, climb up the stairs, or lift heavy objects)1–3,14,15,19. Muscle loss is a common feature found in patients diagnosed with cancer, independent of the specific cancer type. Muscle depletion rates exceeding 50% have been observed in advanced cancer patients, especially those with pancreatic, lung, and gastrointestinal cancers, and are strongly associated with cancer cachexia. Importantly, muscle depletion may occur independently of body weight loss or a drop in body mass index. For this reason, changes in muscle mass should be identified in addition to changes in body weight1–3,17, especially as muscle depletion can be hidden in patients with excess body weight (i.e., overweight or obese) at the time of diagnosis12,13.
Although there is no specific instrument for Oncology, tools are validated for other contexts capable of assessing muscle function, such as the ones proposed in the present protocol (Supplementary data IV).
Managing the patient with or at risk of malnutrition and/or muscle depletion
After screening patients with the proposed protocol, the practical conduct of the clinician according to the level of risk encountered in the patient is presented in (Fig. 1). Fig. 2 represents the nutritional care process activated by the screening process (Supplementary data I for tools).
Identification of patients with or at risk of malnutrition and/or muscle depletion should ideally be followed by referral to appropriate multidisciplinary care team members for full nutrition and strength/mobility assessments to guide a tailored therapy intervention plan27,28,55.
However, as the availability of professionals and time limitations might hinder such actions, our protocol also guides managing and monitoring patients during treatment (Fig. 2). The ESPEN recommendations for actions also link to further evidence-based nutrition resources for healthcare professionals and patients56.
Patients with no nutritional risk should be recommended to monitor their weight. In addition, these patients should receive basic dietary counseling to maintain or improve their nutrient intake during treatment. Physical activity should be encouraged. It has many benefits, including improved muscle strength, reduced fatigue and anxiety, and improved quality of life56 (Fig. 2). Patients reporting no change in strength or mobility should be advised to monitor their activity levels and engage in regular physical activity to reduce the risk of atrophy.
Early nutritional support for patients identified with moderate or high risk of malnutrition or muscle depletion can reduce the possibility of therapy-threatening adverse events and optimize the likelihood of treatment success and long-term survival. Early nutritional intervention may be associated with improved outcomes and a better quality of life, including emotional and psychological status. Additional studies highlight the benefits of early and prospective nutritional management during systemic antineoplastic therapy. Specifically, studies have shown the benefits of oral nutritional supplements (ONS) and enteral nutrition for patients undergoing antineoplastic treatment57–60.
When a clinical nutritionist is part of a multidisciplinary oncology team, best practice involves the immediate and direct referral of patients who are malnourished or at risk of malnutrition, or who have low strength or mobility, for further assessment. However, the expert panel recognized that referral for a clinical nutritionist is not always available, and the treating oncologist may be required to advise and ensure that patients have access to nutritional supplementation, as required61. Globally, in most developed countries, nutrition products are reimbursed by the state for patients. In Portugal, this became a partial reality in 2025. The Portaria No. 82/2025/139, of March 4, establishes the exceptional reimbursement scheme for health technologies related to enteral nutrition, covering enteral formulations, modular formulations, and ONS, which will take effect from August 1, 2025.”
The ESPEN and the European Society for Medical Oncology provide detailed guidance on nutritional support and intervention for patients diagnosed with cancer and undergoing antineoplastic treatment31,56.
These guidelines provide recommendations for total daily energy that should meet the standard daily energy expenditure of healthy adults of between 25 and 30 kcal/kg/day, protein intake of > 1.2 g/kg/day, and, if possible, up to 1.5 g/kg/day, and vitamin and mineral supplementation equal to the recommended daily amounts for healthy individuals or to correct any diagnosed deficit. The source of protein is a relevant factor for muscle anabolism. The source of protein should also be considered for optimal muscle anabolism4,21,62.
Energy requirements might be achieved with the usual recommendations of “little and often” and “fortified foods” or with ONS, which can also improve protein and micronutrient intake. Furthermore, enteral nutrition must be considered if oral intake remains inadequate to meet requirements despite added ONS. Guidelines also recommend maintenance or an increased level of physical activity in patients with cancer to support muscle mass, strength/mobility, and metabolic pattern (ESPEN)56,61,63.
Physical activity and individualized resistance exercise can support health-related quality of life and self-esteem, as well as a reduction in fatigue and anxiety for patients with cancer, and should be encouraged to reduce risks of atrophy due to inactivity (ESPEN)56.
When should nutritional checks be undertaken in patient consultations, and by whom?
The expert panel suggests that early identification of nutritional risk should be undertaken at diagnosis, regardless of age, disease stage, or cancer type (Fig. 1). As such, checkpoints for identification of nutritional risk should be undertaken at any scheduled or unscheduled medical oncology/radiation oncology visit. Nutritional monitoring and assessment of nutritional risk should be conducted at each treatment cycle, monthly, or as clinically indicated, to ensure timely identification and management of nutritional decline during cancer therapy. The panel recommended that checkpoints for the identification of nutritional risk should be performed at the time of disease recurrence and whenever a treatment change is considered before and after surgery2,4,64,65.
The expert panel endorses that the initial identification of nutritional risk should be undertaken by the medical or radiation oncologist and/or the oncology nurse, clinical nutritionist (when available), or other attending healthcare professional leading patient care. Some specialist centers may have a Nutrition Service to which patients are automatically referred for nutritional evaluation and support at diagnosis. However, for most patients, their primary interaction will be with their treating physician, which is the reason for the recommendation. Ideally, patients identified at regular checkpoints and via monitoring of nutritional status during antineoplastic treatment can then be referred for a detailed nutritional assessment by nutrition experts. There was consensus that it is not expected that medical oncology specialists undertake complete nutritional assessments, unless if fully trained and scientifically differentiated in clinical nutrition, according to the recently created competence in Clinical Nutrition of the National Board of Physicians (Ordem dos Médicos), with already more than 50 physicians experts in clinical nutrition. Thus, given the national reality, if a referral is not possible, early identification of malnutrition and muscle depletion should be a key point for management by members of the multidisciplinary team31,32,56.
Conclusion
Malnutrition and/or muscle depletion significantly impact cancer patients and, consequently, their prognosis. State-of-the-art cancer care involves a multidisciplinary and multimodal intervention supported by the best evidence and clinical practice recommendations. Timely and systematic screening for malnutrition and muscle depletion, using validated tools, allows the identification and stratification of nutritional risk and, consequently, structuring the most appropriate needs and interventions.
The proposed protocol aims to streamline the integration of key issues for nutritional management and treatment of cancer patients in a comprehensive and adapted manner to various clinical scenarios. To achieve this, we propose identifying and stratifying nutritional risk and muscle depletion in oncology for a prompt approach and intervention by healthcare professionals. This approach should be multidisciplinary throughout the patient’s journey to ensure continuity of care and the best outcomes. Therefore, it is recommended that physicians, nurses, pharmacists, nutritionists, and other healthcare professionals working in the field of oncology be vigilant for signs and symptoms with nutritional impact.
For most patients, the focus should be identifying those at increased risk of malnutrition and/or muscle depletion at the time of diagnosis and monitoring for any change in status throughout treatment. Nutritional support should be valued as an essential component of the management of all patients with cancer undergoing antineoplastic therapy. In the same way, oncologists routinely evaluate a range of factors, including standard considerations such as blood counts and organ function, to determine whether a patient can commence antineoplastic treatment.
Where possible, delays in initiating antineoplastic therapy due to poor nutritional status should be avoided via the early identification of nutritional status and patient risk. However, the need to initiate immediate antineoplastic therapy must be considered alongside the risk of treatment-related toxicities and early treatment discontinuation in those whose nutritional status is poor. To prevent this nutritional decline, a standardized approach to identify and monitor the nutritional risk of patients commencing and undergoing antineoplastic therapy is proposed with the present protocol, which is feasible within the context of a demanding oncology practice.
Nutritional care has to be aligned with nutritional screening, both integrated with the oncological diagnosis and the initiation of medical therapy. Based on the most recent evidence, early identification of patients at risk or already malnourished during antineoplastic therapy is crucial to optimize treatment success. However, the need does not deflate on malnutrition screening; hence, frailty, muscle mass depletion, and muscle function are markers of early nutritional and clinical impairment that should be priority parameters for clinical assessment of all patients. Depletion of these variables predicts that the patient may not tolerate the recommended treatment intensity and duration, potentially impacting its effectiveness and short- and long-term outcomes.
Looking ahead in the field of medicine and nutrition, the proposed protocol offers more than just clinical guidance; it lays the groundwork for innovation through data integration. Designed to be simple, efficient, and applicable at the start of any anticancer therapy regardless of type or stage, the protocol enables continuous monitoring and early detection of nutritional issues. Building on this vision, the Centre for Interdisciplinary Research in Health and Católica Medical School are already developing a machine learning model that incorporates the full spectrum of patient data. This model aims to enhance diagnostic accuracy and personalize nutrition care across healthcare institutions, making proactive, data-driven interventions a reality in oncology.
Acknowledgments
We gratefully acknowledge S. Torcato-Parreina, RN for her assistance in designing the study and for her expert advice.
Supplementary data
Supplementary data are available at DOI: 10.24875/RPO.25000015. These data are provided by the corresponding author and published online for the benefit of the reader. The contents of supplementary data are the sole responsibility of the authors.
Author contributions
All authors contributed to the conception, design, writing, and approval of the protocol presented in the manuscript. The opinions and recommendations presented here are those of the authors only.
Funding
Project with the support of Nestlé Health Science Portugal.
Conflicts of interest
None.
Ethical considerations
Protection of humans and animals. The authors declare that no experiments involving humans or animals were conducted for this research.
Confidentiality, informed consent, and ethical approval. The study does not involve patient personal data nor requires ethical approval. The SAGER guidelines do not apply.
Declaration on the use of artificial intelligence. The authors declare that no generative artificial intelligence was used in the writing of this m